Empirical Formula of Butane
CH 3 CH 2 12 CO 2 H. Glucose Molecular Formula and Facts.
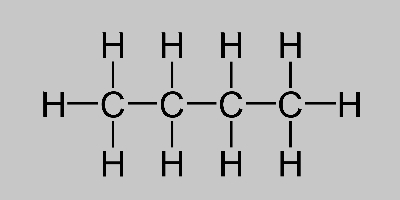
Butane Formula Learn About Butane Isomers And Its Structure
Indeed three gauche butane interactions may be identified in each of the cis-decalin conformations as will be displayed by clicking on the above conformational diagram.
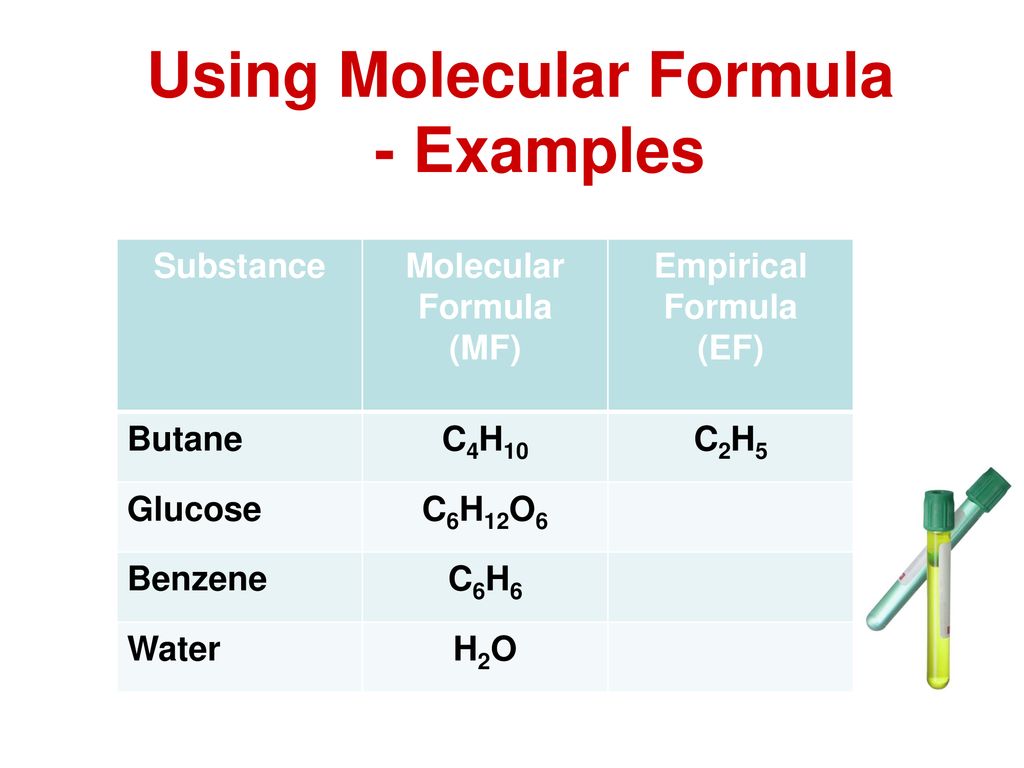
. The units for Θ dont match since the this is an empirical equation - a result of experience and experiments. Common Functional Groups in Organic. Benzene one of the simplest aromatic hydrocarbons serves as the raw material for the synthesis of many synthetic drugs.
This provides support for the empirical. To maintain the water temperature - heat must be supplied to the water. Furthermore hydrocarbons such as propane and butane are used for commercial fuel purposes in the form of Liquefied Petroleum Gas LPG.
Unlike other chemical formula types which have a limited. The structural formula of a chemical compound is a graphic representation of the molecular structure determined by structural chemistry methods showing how the atoms are possibly arranged in the real three-dimensional spaceThe chemical bonding within the molecule is also shown either explicitly or implicitly. Properties of Ionic and Covalent Compounds.
CH 3 CH 2 10 CO 2 H. E943A R-600 E-943A E 943A Diethyl N-C4H10 N-Butane Normal-Butane C4H10 Molar Mass C4H10 Oxidation Number Dioxygen - O 2 Lox O₂ Oxygen Oxygen Gas Liquid Oxygen Triplet Oxygen Diatomic Oxygen Molecular Oxygen O2 Bond Polarity. Ethanol Molecular Formula and Empirical Formula.
Due to their different molecular structures the empirical formula of hydrocarbons. Education for Ministry EfM is a unique four-year distance learning certificate program in theological education based upon small-group study and practice. Required heat to cover evaporation can be calculated as.
Most of the heat or energy required for the evaporation is taken from the water itself. Pentan-1-ol pentan-2-ol and pentan-3-ol are structural isomers that exhibit position isomerism. Butane and isobutane C 4 H 10 are structural isomers of each other.

Chem C10 Empirical Molecular Formula Flashcards Quizlet

Butane Formula Structure What Is Butane Used For Video Lesson Transcript Study Com

Empirical Formula Of A Compound Ppt Download

Butane Molecular Geometry Hybridization Molecular Weight Molecular Formula Cas Number Bond Pairs Lone Pairs Lewis Structure
0 Response to "Empirical Formula of Butane"
Post a Comment